The key role of microbes in nitrogen fixation. How overuse of nitrogen-containing fertilizers can cause algal blooms. Micro-organisms are necessary for decomposing animal and plant matter in the nitrogen cycle. The nitrogen cycle is the circulation of nitrogen in various forms through nature. Learn more about this process, which is critical to the survival of plants and animals. Nitrogen fixation, in which nitrogen gas is converted into inorganic nitrogen compounds, is mostly (90 percent) accomplished by certain bacteria and blue-green algae. The cycle itself is a complex biochemical system where nitrogen is changed from its natural molecule N2 into a form that is useful for the biological process. Most life can not use nitrogen in its natural form.
The Nitrogen Cycle The nitrogen cycle explains the how nitrogen flows between animals, bacteria, plants, the atmosphere, and the soil on earth. The uniqueness of the nitrogen cycle is that nitrogen is the most abundant gas in the earth’s atmosphere, about 78% of all air, but it can’t be directly utilized by the animals and plants unless it is converted into usable compounds. Its importance is because of its key role in the formation of nucleic and amino acids.
It is also an essential part of adenosine triphosphate (ATP), which is the chief energy molecule for living things. For nitrogen to be used by plants and animals, it has to change into various states through the nitrogen cycle. The major changes in the nitrogen cycle include nitrogen fixation, nitrification, assimilation, ammonification, and denitrification. These changes to different nitrogen oxides are dependent on various activities of microorganisms such as bacteria and fungi. Processes of the Nitrogen Cycle • Nitrogen Fixation Nitrogen fixation is the process of converting the atmospheric nitrogen (N 2) into biological state nitrogen. It is the first process of making nitrogen available for plants. It is defined as an anaerobic (without oxygen) process that catalyzes the reduction of atmospheric nitrogen (N 2) into ammonia (NH 3).
The process is solely carried out by prokaryotes (bacteria) which have the natural strength to break the triple bond between the nitrogen atoms. These nitrogen-fixing organisms are free-living bacteria whereas others are symbiotic nitrogen fixers. An example of a nitrogen fixer is the Rhizobium bacteria in the roots of legumes (soybeans, peas or clovers). Other types of nitrogen-fixing prokaryotes are extensively distributed in different environments including terrestrial and aquatic settings.
A special enzyme known as dinitrogenase is responsible for the fixation process. Once the nitrogen has been reduced to ammonia, the plants can now use it to make other biological compounds through the synthesis of enzymes, nucleic acids, chlorophyll, and proteins. • Nitrification Nitrification is the process where the ammonium ions (NH 4) are converted into nitrides, first into nitrites (NO 2 –) then into nitrate (NO 3 –). Still, this process is done by the nitrogen-fixing bacteria. The first step is the oxidation of ammonia to nitrate, done by microbes termed as ammonia-oxidizers.
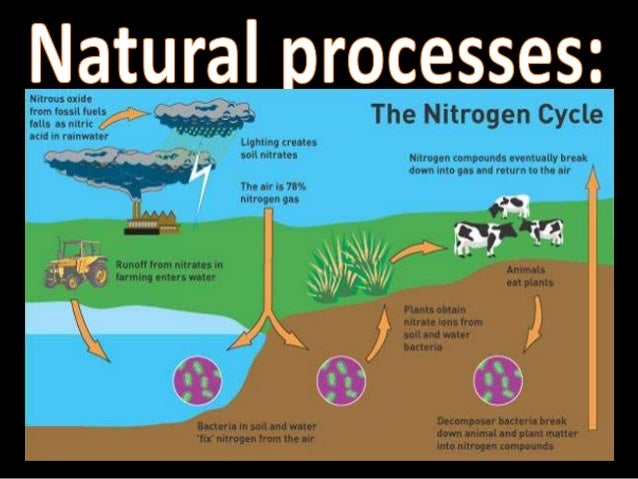
The second step is the oxidation of nitrite (NO 2 –) to nitrate (NO 3 –). The participating bacteria here are termed as nitrogen-oxidizing bacteria and they include nitrococcus, nitrobacters, and nitrosomonas.

• Assimilation Assimilation refers to how plants and animals obtain nitrogen. Plant roots absorb nitrates from the soil into the roots then into the entire plant system. The plants then use the nitrates in the synthesis of nucleic acids, enzymes, amino acids, proteins, and chlorophyll. On the other hand, animals assimilate nitrogen by eating the plants. • Ammonification Ammonification is also termed as the decaying process. It occurs when the plant or animal dies then decomposers such as fungi and bacteria decompose the tissues and transforms the nitrogen back into ammonium.
The Nitrogen Cycle Diagram
The ammonium then reenters the nitrogen cycle where it is taken up by plants and other microorganism for development. Animal waste equally releases ammonium into the nitrogen cycle. • Denitrification Denitrification is the process that changes nitrate to nitrogen gas, hence returning it into the atmosphere. This process releases the excess nitrogen in the soil back into the atmosphere.
The Nitrogen Cycle Could Not Exist Without
Special prokaryotes known as denitrifying bacteria carry out this process of reversing nitrates into nitrogen gas. Unlike nitrification, this process is anaerobic (uses oxygen) and the bacteria involved are in the genus Pseudomonas, Paracoccus, and Bacillus. Dinitrogen gas (N 2) is the final outcome of denitrification, but other nitrogen-related gaseous forms can as well be released. A good example is nitrous oxide (N 2O) which is considered a potential greenhouse gas.
• What is the most common way that nitrogen fixation occurs? • a.lightning • b.nitrogen fixing bacteria • c.fossil fuel combustion • d.forest fires • Atmospheric nitrogen (N 2 gas) is easily taken up and used by plants and animals. • a.true • b.false • Which of the following is a component of acid rain? • a.ammonium • b.denitrifying bacteria • c.nitrogen oxides • d.organic nitrogen • Organic nitrogen is coverted back to inorganic nitrogen like ammonium through the process of • a.nitrogen fixation • b.nitrification • c.decay • d.none of the choices • Eutrophication in coastal waters can cause an increase in • a.respiratory diseases in fish • b.agricultural productivity • c.harmful algal blooms • d.denitrification • Ammonium (NH 4) stays in soil, while nitrate (NO 3) is easily leached out. Why do they behave so differently? • a.NO 3 is broken down by bacteria, so it is easily carried away • b.NH 4has a positive charge and sticks to soil particles. • c.NH 4 is unavailable to organisms, so it remains unchanged in the soil.
 RSS Feed
RSS Feed
